- Retina Research Foundation
- About RRF
- Pilot Study Grants
- Grant Recipients 2024
- Samuel M. Wu, PhD
- Yingbin Fu, PhD
- Rui Chen, PhD
- Graeme Mardon, PhD
- Richard L. Hurwitz, MD
- Lih Kuo, PhD
- Wenbo Zhang, PhD
- Curtis Brandt, PhD
- Timothy Corson, PhD
- Jianhai Du, PhD
- Francesco Giorgianni, PhD
- James Monaghan, PhD
- Milam Brantley, MD, PhD
- Seongjin Seo, PhD
- Vladimir Kefalov, PhD
- Andrius Kazlauskas, PhD
- Erika D. Eggers, PhD
- Ann C. Morris, PhD
- Ming Zhang, MD, PhD
- Christine Sorenson, PhD
- Alex J. Smith, PhD
- Jeffrey M. Gross, PhD
- David M. Wu, MD, PhD
- Kinga Bujakowska, PhD
- Eric Weh, PhD
- Ching-Kang Jason Chen, PhD
- Jakub K. Famulski, PhD
- Thanh Hoang, PhD
- Georgia Zarkada, MD, PhD
- Eleftherios Paschalis Ilios, PhD
- Publications
- Grant Guidelines and Information
- Grant Application
- Grant Recipients 2024
- Research Programs
- Contact Us
- Giving
- RRF History
- Home
Matthews Professorship
M D Matthews Professor

Bikash Pattnaik, PhD
Department of Pediatrics, Ophthalmology & Visual Sciences
University of Wisconsin
McPherson Eye Research Institute
Madison, WI
Current Research Interests
Vision loss due to ion-channelopathy
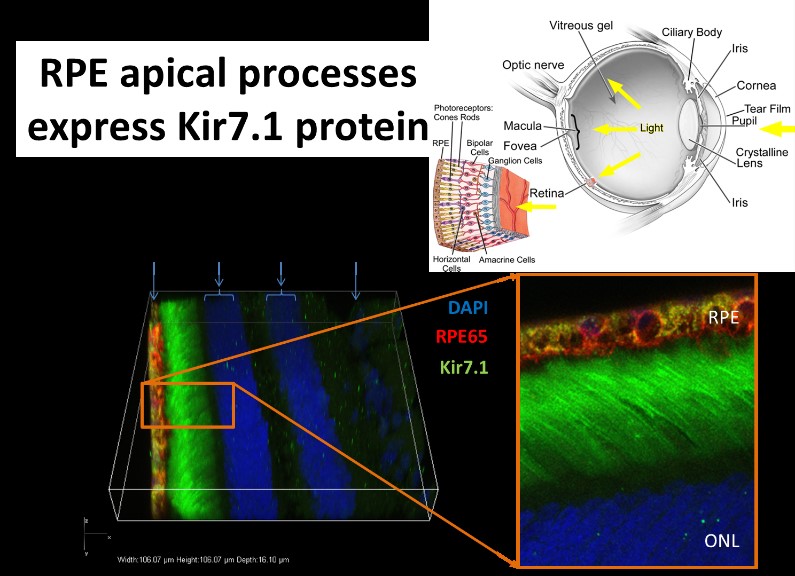
Genetic eye diseases stand out as the leading cause of blindness across all ages, from infants to adults. Dr. Pattnaik’s research focus is the use of both forward and reverse genetics to determine the genetic basis of blindness due to mutations in an inwardly rectifying potassium channel (Kir7.1). This protein is present in the retinal pigment epithelium (RPE) within the retina that helps with the diffusion of potassium across the cell. In both induced pluripotent stem cells (iPSC) derived RPE cells from a Leber Congenital Amaurosis patient and in mice with Kir7.1 knock-down, cellular function was altered to cause blindness. Dr. Pattnaik’s lab has recently developed gene-therapy treatment for patients that is in an advance stage clinical translation through Hubble Therapeutics. Using a particular nonsense mutation disease model, Pattnaik’s lab is pursuing small molecule drugs, or biological molecules such as DNA or RNA that can be targeted to RPE cells as other possible treatments for pediatric blindness.
Ion channelopathies are diseases caused by defective ion channels. Ion channels are membrane proteins distributed throughout the human body and mediate cell-cell communication through passage of specific ions into or out of cells. Patients with a specific potassium channel gene (KCNJ13, encoding Kir7.1 protein present in the Retinal Pigment Epithelium RPE) coding variants are diagnosed with a spectrum of blindness phenotypes: Snowflake Vitreoretinal Degeneration (SVD) and Lebers congenital Amaurosis (LCA). Dr. Pattnaik has shown that a loss-of-function in the Kir7.1 channel disrupts RPE and photoreceptor cell communication using ectopic expressions and a mouse model that demonstrate pathological features in carriers.
RPE cells are retina glial cells that support our ability to see. Function of these RPE cells relies on the activity of ion channels and transporters confined to the membrane micro domain. Dr. Pattnaik is using small animal models, cell culture system and patient derived iPSC-RPE cells to understand cellular phenotypes. Any identifiable distinction between the control and diseased cells permit us to test efficacy of various therapeutic measures through “patient-in-a-dish” approach. Some of the experimental strategies his laboratory currently uses are small molecule drugs to overcome translational errors, or use of gene manipulation to measure Kir7.1 current through patch-clamp electrophysiology outcome.
Dr. Pattnaik’s future focus is on the basic understanding of the cellular basis of potassium and chloride channel function in the retina as tools to diagnose disease so as to develop its prevention.